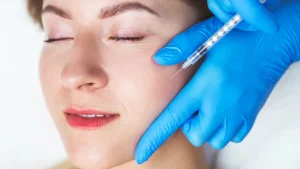
A new name is making waves in the beauty industry – today we’ll talk about the NEAUVIA™ filler brand.
NEAUVIA™ offers a spectrum of dermal fillers meticulously designed to help with various signs of aging. We have seen some fantastic transformations after NEAUVIA™ injection.
However, don’t worry, this isn’t going to be just another incomplete article in a blog, focusing only on benefits and positive aspects. Medica Depot is here to provide product and treatment information in more ways than one – including potential side effects, contraindications, and recovery timelines. Let’s dive in!
What is NEAUVIA™ Dermal Filler?
NEAUVIA™ is an organic line of dermal fillers formulated to rejuvenate and enhance the skin’s appearance. The primary ingredient in NEAUVIA™ products is hyaluronic acid, which operates as a humectant, attracting and retaining water molecules to bolster the skin’s moisture levels.
What patients like the most is the natural look they get, which is different from other aesthetic interventions. Dermal fillers are used to address common signs of aging but also to alleviate osteoarthritis symptoms since they’re a natural component found in the synovial fluid of joints, acting as a lubricant and shock absorber.
Methods of Application
NEAUVIA™ organic filler is typically administered through a series of injections. The injection techniques depend on the specific NEAUVIA™ product you are using and the area being treated.
You should start with an in-depth evaluation of the patient’s facial anatomy, a decisive step for a tailored and effective treatment. After that, if you have not observed any contraindications or potential problems, you can proceed.
NEAUVIA™ Aftercare
While the majority of individuals can resume their normal activities shortly after the procedure, providing them with proper aftercare tips is very important.
The following tips can promote healing and maintain the longevity of the results:
- Immediate post-treatment period: Applying a cold compress intermittently in the first 24 hours can help alleviate swelling and discomfort.
- Avoiding strenuous activities: It is advisable to refrain from engaging in strenuous physical activities, including vigorous exercise and sauna sessions, for at least 24 to 48 hours post-treatment.
- Gentle skincare routine: Avoiding abrasive scrubs or harsh skincare products can prevent irritation to the treated areas.
- Limiting sun exposure: Patients should use sunblock with a high SPF on treated areas and consider wearing a hat or using other protective measures when outdoors.
- Avoiding alcohol and blood thinners: Abstaining from alcohol consumption and avoiding blood-thinning medications or supplements is crucial, as these can contribute to increased bruising.
How Long Does NEAUVIA™ Fillers Last?
The duration of NEAUVIA™ is anywhere from 12 to 24 months, which is why these fillers are famous for their enduring effects. The unique formulation of NEAUVIA™, which incorporates hyaluronic acid and polyethylene glycols (PEGs), contributes to the sustained presence of the filler beneath the skin.
The incorporation of PEGs further supports the stability of the filler, prolonging its effectiveness. It’s important to note that individual variations, such as metabolism and skin characteristics, can influence the duration of the results. You should inform your clients about that to prevent dissatisfaction and complaints in the future.
Potential Side Effects and Contraindications
Side effects of hyaluronic acid are usually negligible, but it’s best to explain them to your patients upfront. The most common side effects are:
- Temporary swelling and redness: Mild swelling and redness at the injection site are normal and usually resolve within a few days.
- Bruising: Typically temporary and can be easily concealed with makeup.
Certain conditions may contraindicate the use of these dermal fillers, including allergies to PEGs and vitamin C since there are NEAUVIA™ with vitamin C. Individuals with a history of severe autoimmune disorders or certain skin conditions may not be suitable candidates.
Is NEAUVIA™ Filler FDA-approved?
No, NEAUVIA™ fillers are not officially FDA-approved, but that should not worry you. This in no way indicates that they are unsafe or that your patients may have any problems. The absorbable materials used in NEAUVIA™ (mainly hyaluronic acid) are FDA-approved, which is a great indicator of safety.
On top of that, NEAUVIA™ devices, such as Plasma IQ and LaserME, are FDA-approved, which shows the trustworthiness of this company.
The Verdict: NEAUVIA™’s Role in Facial Rejuvenation
NEAUVIA™ organic fillers represent a cutting-edge solution for those seeking a non-invasive method of facial rejuvenation. NEAUVIA™ has indisputably earned its status as a revered name in the vast landscape of cosmetic products.
While reveling in the positive attributes of NEAUVIA™, it is of utmost importance to underscore the significance of an individual approach to each client. This approach ensures that the experience is not only personalized but also exemplifies the highest standards of safety and well-being.
FAQ
Is NEAUVIA™ a good filler?
NEAUVIA™ has gained recognition as a reputable dermal filler brand in the field of aesthetic medicine. Known for its focus on natural-looking results, longevity, and versatility, NEAUVIA™ is considered by many as a good option for those seeking non-surgical cosmetic enhancements.
What is NEAUVIA™ used for?
NEAUVIA™ dermal fillers are primarily used to address common signs of aging, such as wrinkles, marionette lines, nasolabial folds, fine lines, glabellar lines, and loss of facial volume. The versatile product line allows for customized treatment approaches, targeting specific areas.
Where is NEAUVIA™ filler from?
NEAUVIA™ is a dermal filler brand that originates from Europe, with main locations in Switzerland (Geneva), Italy (Brindisi), and Poland (Warsaw). Produced under stringent quality standards, NEAUVIA™ products are developed to meet the highest regulatory requirements.
References
- Jang HJ, Shin CY, Kim KB. Safety Evaluation of Polyethylene Glycol (PEG) Compounds for Cosmetic Use. Toxicol Res. 2015 Jun;31(2):105-36. doi: 10.5487/TR.2015.31.2.105. PMID: 26191379; PMCID: PMC4505343.
- Kubik P, Gallo D, Tanda ML, Jankau J, Rauso R, Gruszczyński W, Pawłowska A, Chrapczyński P, Malinowski M, Grzanka D, Smolińska M, Antosik P, Piesiaków ML, Łukasik B, Pawłowska-Kubik A, Stabile G, Guida S, Kodłubański Ł, Decates T, Zerbinati N. Evaluation of the Safety of NEAUVIA™ Stimulate Injectable Product in Patients with Autoimmune Thyroid Diseases Based on Histopathological Examinations and Retrospective Analysis of Medical Records. Gels. 2023 May 26;9(6):440. doi: 10.3390/gels9060440. PMID: 37367111; PMCID: PMC10297656.
- Zerbinati N, Mocchi R, Galadari H, Maccario C, Maggi M, Rauso R, Passi A, Esposito C, Sommatis S. In Vitro Evaluation of the Biological Availability of Hyaluronic Acid Polyethylene Glycols-Cross-Linked Hydrogels to Bovine Testes Hyaluronidase. Biomed Res Int. 2019 Jun 12;2019:3196723. doi: 10.1155/2019/3196723. PMID: 31309104; PMCID: PMC6594335.