The 2019 Global Burden of Disease study focused on the demographics of osteoarthritis patients. It revealed that 73% of those affected are aged 55 and above and that 60% of these individuals are female. This emphasized the prevalence of osteoarthritis, marking it as a common joint disease in this age demographic.
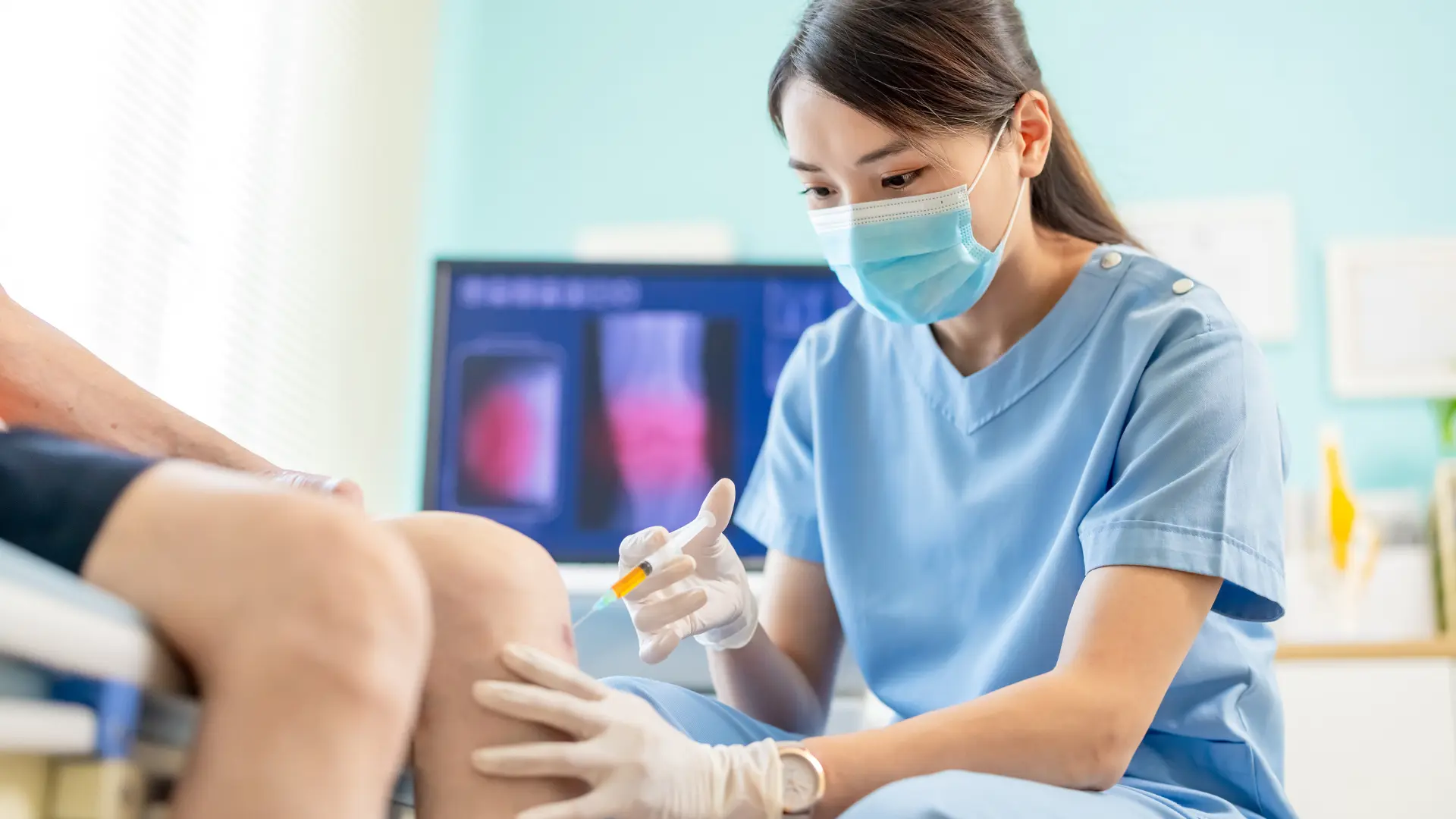
Osteoarthritis causes pain, swelling, and stiffness in the specific joint. It often restricts individuals from carrying out their daily activities due to the discomfort. While this lifelong condition remains incurable, Durolane presents a minimally invasive treatment option. This single-injection therapy delivers temporary symptomatic relief, allowing patients to enhance joint functionality and quality of life.
In this article, we will explore Durolane and how it works, its manufacturing process, and the company behind this single-injection therapy for knee osteoarthritis.
Key Takeaways
- The Durolane manufacturer dedicates itself to delivering clinically proven and cost-effective products that can quickly and safely address and successfully treat individuals.
- Bioventus is relentless in pursuing scientific excellence and patient-centric innovation.
- With the help of NASHA technology, the stabilized HA formulation in Durolane slows down degradation for a longer duration of effect.
- The FDA has approved Durolane to treat knee osteoarthritis pain in individuals who have failed to respond to therapies and oral medications.
- Bioventus, LLC, has a solid foundation of market-leading products and compelling growth aspects.
Introduction to the Manufacturer of Durolane
Durolane is a prescription drug used to treat osteoarthritis symptoms. The manufacturer, Bioventus, LLC, produced the treatment as a single-injection therapy. The injection contains a high concentration of hyaluronic acid to lubricate and cushion the knee joints.
Bioventus remains committed to revolutionizing the treatment of OA and joint-related conditions. In 2019, Bioventus entered a development collaboration agreement with MTF Biologics to co-develop a next-generation placental tissue product for knee OA. This also aims to develop additional uses and indications beyond the focus of knee OA as part of their partnership.
The Durolane manufacturer dedicates itself to delivering clinically proven and cost-effective products that can quickly and safely address and successfully treat individuals. The orthobiologic products focus on osteoarthritis and surgical and non-surgical bone healing. In line with research and development, the company acquired CartiHeal in 2021, a transformational technology in knee osteoarthritis treatment.
The Durolane manufacturer is relentless in pursuing scientific excellence and patient-centric innovation. It leads the innovations of active healing and orthobiologics, fronting clinically productive and cost-effective solutions for medical practitioners and patients. Its patient-centric approach allows it to meet patients’ specific needs and improve their quality of life.
The Science Behind Durolane

Durolane’s unique formulation offers a single-injection treatment for symptomatic relief from osteoarthritis. It utilizes the non-animal stabilized hyaluronic acid (NASHA) technology, creating a highly concentrated and stabilized form of HA.
This injection treatment can address the patient’s pain, swelling, and knee joint stiffness concerns. Durolane can offer a practical and long-lasting solution for osteoarthritis. Its single-injection therapy provides convenience and a less invasive alternative to multiple injection procedures.
So, what is Durolane made of? This treatment relies heavily on its primary ingredient, hyaluronic acid. This substance is a crucial component of the synovial fluid that lubricates the joint. With the help of NASHA technology, the stabilized HA formulation in Durolane slows down degradation. It allows the HA to remain in the joint for a longer duration.
Aside from HA, a few additional Durolane ingredients back its formulation to offer effective results to individuals.
- Sodium Chloride: This is the phosphate-buffered saline in the Durolane formulation, suspending sodium hyaluronate. Sodium chloride aids in maintaining the formulation’s isotonicity, imitating the environment within the body cells.
- Potassium Dihydrogen Phosphate: A soluble salt frequently helps maintain a stable pH in buffer solutions. This helps in preserving the Durolane injection solution’s stability and pH.
- Disodium Hydrogen Phosphate Dihydrate: This additional ingredient works with potassium dihydrogen phosphate to maintain the pH in the Durolane formulation. This combination ensures the pH’s stability and compatibility with the body’s cells.
The hyaluronic acid (HA) in Durolane’s formulation works as a natural lubricant and shock absorber. In osteoarthritis, the HA naturally produced in the joints decreases over time. Fortunately, Durolane replenishes the joint’s HA to restore its ability to function efficiently without pain.
When does Durolane work? Patients can notice positive effects a few days after the treatment. They can fully reach optimal results after a week or two. Durolane injections can last six (6) months or longer as they restore lubrication and shock absorption. Medical professionals may also recommend subsequent sessions to maintain the therapy’s relieving effect.
- Direct Treatment: The Durolane injection can address and target the affected joint, providing precise relief. Medical professionals can effectively treat the specific joint to deliver symptomatic relief.
- Repeatable Treatment: Medical professionals may require repeated treatment, allowing for longer-lasting outcomes and ongoing management of symptoms.
- Delaying Surgery: Undergoing a Durolane injection therapy may help avoid or delay invasive procedures like replacement surgeries.
- Long-Term Relief: This single-injection therapy can provide long-term freedom from osteoarthritis symptoms such as pain, swelling, and stiffness.
- Lubrication and Cushioning: HA is a natural substance in synovial fluid in joints. When Durolane refills this essential compound, it provides lubrication and cushioning in the joint.
- Reduced Friction and Inflammation: When medical practitioners directly inject Durolane into the joint, it can minimize friction and inflammation. Durolane efficiently decreases the occurring OA symptoms.
- Delay in Knee Replacement: A 2015 clinical study has shown that HA treatments can substantially delay the need for knee replacement surgery.
Manufacturing Process of Durolane
In a Durolane vs. Orthovisc comparison, here is some information on how the former differs. Durolane offers a clear and viscous gel backed by the potent hyaluronic acid. The Durolane manufacturer biosynthesized sodium hyaluronate using bacterial fermentation and NASHA technology to stabilize naturally entangled HA chains to produce a gel bead-like structure.
It’s a single injection, single dose preparation, and only allowed for single use. Professionals must store the syringe appropriately, and physicians should use the contents immediately after opening the packaging. The Food and Drug Administration (FDA) states that medical practitioners must follow strict aseptic administration. Moreover, they should properly dispose of the Durolane syringe after use.
The FDA has approved Durolane to treat knee osteoarthritis pain in individuals who have failed to respond to therapies and oral medications. Additionally, medical practitioners can use it for symptomatic treatment associated with mild to moderate joint OA pain, which includes the knee, ankle, shoulder, elbow, wrist, fingers, and toes.
Durolane may not have a long list of contraindications, but the treatment has not been tested on breastfeeding or pregnant women and children (21 years or younger). Moreover, individuals who experience the following should avoid undergoing a Durolane injection treatment:
- Patients with knee joint infections, infections, or skin disease in the area of the injection site.
- Patients with known hypersensitivity or allergy to hyaluronic acid preparations.
Durolane also utilizes advanced technologies and state-of-the-art components, which completes this effective injection treatment.
- NASHA Technology: The Durolane manufacturer uses this technology to stabilize the entangled hyaluronic acid chains to create a clear and dense gel. Durolane uses highly purified and high-molecular-weight HA in the injections, allowing optimal treatment efficacy.
- Single-Injection: Durolane boasts its single-injection therapy for knee OA. It uses a lightly cross-linked and highest molecular weight HA among all US-approved HA products. Durolane also has more Level 1 clinical studies than other single-injection OA.
- Longer-Lasting Relief: Compared to NSAIDs and other therapies, Durolane offers more extended relief from OA symptoms. It is also safe for repeated treatments, as it does not increase the incidence of adverse effects.
Clinical Evidence Supporting Durolane
According to a clinical evidence study in 2018, the assessment of Durolane and NASHA technology effectively relieved knee OA symptoms. Single-injection NASHA benefits the clinical management of knee osteoarthritis. It also shows an excellent biocompatibility profile for NASHA.
Furthermore, an open-label study revealed that Durolane viscosupplementation using NASHA is effective and well-tolerated in treating the symptoms. The study also reported no severe or allergic reactions in the patients injected with Durolane.
A few patients shared their testimonials after their Durolane injection treatments. This procedure helped three physically active patients return to their usual activities. Durolane gave the patients symptomatic relief from painful knee pain and joint swelling.
- An individual used to run races and marathons, but it was not until a bad sprain led to a swollen ankle. MRI confirmed the degenerative joint disease. He talked to his doctor about Durolane and received his first injection afterward. After a few days, the positive result became noticeable, and a few months in, he returned to running.
- The following individual tore his ACL way back, and his doctor advised him that he would most likely get arthritis. Years after, his knee locked up, and an MRI revealed that a knee OA had been starting. His surgeon recommended a Durolane injection. A week later, the patient felt better and returned to his active life.
- Another patient who had an active lifestyle also faced troubles after being diagnosed with osteoarthritis. Interestingly, their family doctor suggested a hyaluronic acid injection to lubricate the joints. Over a decade later, the patient shared that Durolane stabilized the progression of knee OA with no pain or inflammation.
Durolane injections have significantly impacted the daily lives of individuals dealing with OA. The therapy relieves joint pain, stiffness, swelling, and instability. After injections, patients regain control over their lives and return to their usual activities.
Improved quality of life and slowed the progression of OA, avoiding or postponing the possibility of surgery. Moreover, the safety and efficacy of Durolane can empower individuals to achieve relief and return to their routine without discomfort.
Ongoing Research and Future Directions

Bioventus, LLC, has a solid foundation of market-leading products and compelling growth aspects. It has actively acquired technology and collaborated with large companies to further innovate in osteoarthritis and orthobiologics. Starting this year, Durolane agrees to a contract with Aetna Medicare Advantage for its plan members to access Durolane injection treatments.
Moreover, Durolane uses NASHA technology to stabilize the HA chains and produce a gel bead-like texture, allowing for an extended duration of symptomatic relief. With Bioventus’ continuous research and development efforts in innovating knee OA relief and cure, the future of knee OA solutions looks promising. Medical professionals must administer the treatment to ensure the safety and efficacy of Durolane’s procedure.
While further research and direction information about Bioventus is not readily available online, it’s safe to say that they aim to relieve knee OA symptoms after a single injection and innovate more with this focus. The Durolane manufacturer offers a solution to enhance the quality of life of individuals, allowing them to avoid the discomfort and pain caused by OA.
Conclusion
The company behind Durolane, Bioventus, deserves recognition for its contributions and achievements to the orthopedic field. The development of Durolane for OA symptoms has shown the company’s innovation, research, and patient care. They created a unique formulation using NASHA technology to provide a longer-lasting effect, improved joint function, and a better quality of life.
By ensuring safety for repeated treatments, Bioventus demonstrates its commitment to a patient-centric approach, which can potentially delay future surgical procedures. Bioventus has paved the way for a brighter future in orthopedic medicine and for patients suffering from OA. Ongoing research and developments can promise improved treatments and quality of life for patients and the field.
About: Medica Depot is your trusted all-in-one supplier, offering a range of high-quality medical injectables and supplies. We offer a worry-free experience in searching for the best and most popular products on the market, like Durolane. Whether for health professionals, plastic surgeons, dermatologists, licensed estheticians, or other specialists, we can offer genuine, brand-name products you may need. With Medica Depot, we prioritize serving you better to improve the patient’s quality of life.
References
- Institute for Health Metrics and Evaluation. (2019). GBD 2019: Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Institute for Health Metrics and Evaluation; University of Washington. https://vizhub.healthdata.org/gbd-results/
- DUROLANE ® INSTRUCTIONS FOR USE. (n.d.). Food and Drug Administration. https://www.accessdata.fda.gov/cdrh_docs/pdf17/P170007D.pdf