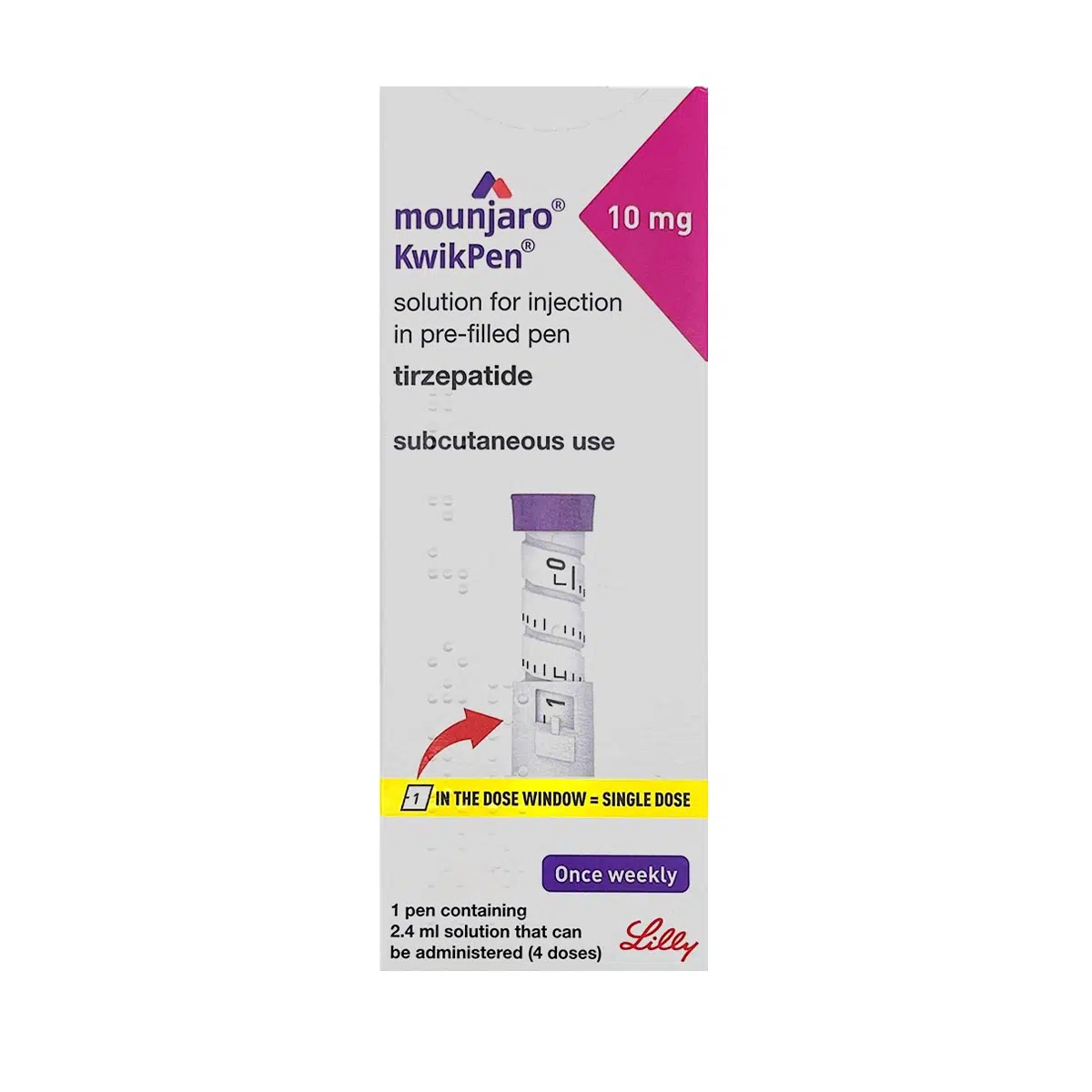
MOUNJARO® 10mg KwikPen®, 1 Stk.
All products are 100% genuine and sourced from trusted manufacturers, with authenticity that can be verified for complete peace of mind.
Order exactly what you need with no minimum quantity required, whether it’s a single unit or a larger order.
Your order is guaranteed to arrive safely, with full shipment tracking and dedicated support to ensure a smooth delivery experience.
Your payments are protected with industry-standard encryption and trusted payment providers, ensuring every transaction is safe, private, and secure.
Manufacturer: Eli Lilly and Company Limited
Active Substance(s): TIRZEPATIDE
Strength: 10mg/0.6ml injection
Pack Size: 1 box with 4 pre-filled KwikPens
-
Cold chain shipping available for temperature-sensitive products.
-
Orders are processed within 1–2 business days.
-
Delivery timelines vary by destination and shipping method — view our Shipping Policy for region-specific estimates.
-
Tracking information will be provided once shipped.
-
No order minimum applies.
-
If your shipment is delayed, lost, or arrives damaged, you're covered under our Refund & Replacement Policy.
We accept Visa, Mastercard, and American Express. Apple Pay and Google Pay are available upon request via your account manager.
All transactions are processed through a secure, PCI DSS–compliant system to ensure your data is fully protected.


Why Choose Medica Depot
Shop hundreds of medical products with no minimums and free shipping on large orders.
INFORMATION
What is MOUNJARO® 5mg KwikPen®
MOUNJARO® 5mg KwikPen® is a multi-dose, pre-filled pen designed to deliver 5 mg tirzepatide in 0.6 mL by subcutaneous injection once weekly. This product is indicated for the treatment of adults with type 2 diabetes mellitus and for weight management in patients who meet BMI criteria. The pen is engineered for single-patient use, providing a safe, convenient method of administration. Each device contains 2.4 mL of solution (total of 20 mg tirzepatide) and supplies four fixed doses. The solution is clear, colourless to slightly yellow, and can be injected in the abdomen, thigh, or upper arm.
-
Dose per injection: 5 mg tirzepatide in 0.6 mL
-
Total pen content: 20 mg/2.4 mL (8.33 mg/mL)
-
Number of doses per pen: 4
-
Injection sites: Abdomen, thigh, upper arm
-
Administration: Subcutaneous only
What is the Administration Protocol for MOUNJARO® 5mg KwikPen®
MOUNJARO® 5mg is administered once weekly at a consistent time, with or without meals. The dosing schedule allows for gradual escalation to minimize gastrointestinal adverse reactions.
-
Initiation: 2.5 mg weekly for 4 weeks, then increase to 5 mg
-
Titration: May increase in 2.5 mg increments every ≥4 weeks (up to 15 mg)
-
Maintenance doses: 5 mg, 10 mg, or 15 mg once weekly
-
Missed dose protocol:
-
If ≤4 days late: administer as soon as possible
-
If >4 days late: skip and resume on next scheduled day
-
-
Injection technique:
-
Attach a new sterile needle for each use
-
Dial pen until 1 appears in the window (=0.6 mL)
-
Inject subcutaneously, hold for 5 seconds until dose window shows 0
-
-
Site rotation recommended to avoid local irritation
What are the Indications for MOUNJARO® 5mg KwikPen®
MOUNJARO® is prescribed for glycaemic control in adults with type 2 diabetes mellitus and as an adjunct for weight management. Contraindications and precautions should be carefully considered in clinical use.
-
Indications:
-
Adults with type 2 diabetes as monotherapy or in combination with other antidiabetic agents
-
Adults with BMI ≥30 kg/m² (obesity)
-
Adults with BMI ≥27 to <30 kg/m² with weight-related comorbidities (e.g., hypertension, dyslipidaemia, OSA, cardiovascular disease, prediabetes)
-
-
Contraindications:
-
Hypersensitivity to tirzepatide or excipients
-
-
Precautions:
-
History of pancreatitis
-
Severe gastrointestinal disorders, such as gastroparesis
-
-
Adverse effects:
-
Common: nausea, vomiting, diarrhoea, abdominal pain, constipation
-
Risk of hypoglycaemia when combined with sulphonylurea or insulin
-
Mild to moderate injection-site reactions
-
Occasional gallbladder events
-
Rare: acute pancreatitis, hypersensitivity reactions
-
What is the composition of MOUNJARO® 5mg KwikPen®
The formulation contains tirzepatide as the active pharmaceutical ingredient, with additional excipients to ensure stability and usability.
-
Active substance: Tirzepatide 5 mg/0.6 mL
-
Total content per pen: 20 mg in 2.4 mL
-
Concentration: 8.33 mg/mL
-
Excipients: Disodium hydrogen phosphate heptahydrate, benzyl alcohol (~5.4 mg per dose), glycerol, phenol, sodium chloride, sodium hydroxide, concentrated hydrochloric acid, water for injections
-
Sodium content: <1 mmol sodium per dose (sodium-free)
What Comes in the MOUNJARO® 5mg KwikPen® Package
MOUNJARO® KwikPen® is supplied in carefully prepared packaging, but needles are not included.
-
Presentation: Pre-filled KwikPen® with 2.4 mL (4 doses)
-
Pack sizes: 1 or 3 pens per carton (availability may vary)
-
Needles: Not supplied in the package; must be obtained separately
-
Documentation: Patient Information Leaflet (PIL) and Instructions for Use (IFU) included
Does MOUNJARO® 5mg KwikPen® cause side effects?
Yes, side effects are possible, with gastrointestinal reactions being most common. These effects are usually dose-related and may decrease over time.
-
Very common: Gastrointestinal disorders – nausea, diarrhoea, vomiting, constipation, abdominal pain
-
Hypoglycaemia: Increased risk when combined with sulphonylureas or insulin; severe hypoglycaemia rare (~0.2%)
-
Injection-site reactions: Up to 3–8% of patients, generally mild
-
Gallbladder events: Reported in up to 2% of patients
-
Uncommon but serious: Acute pancreatitis
-
Rare: Severe allergic reactions (e.g., anaphylaxis, angioedema)
-
Laboratory changes: Small increases in amylase and lipase
How should MOUNJARO® 5mg KwikPen® be stored?
Strict storage conditions must be followed to maintain stability and sterility.
-
Unopened pens: Refrigerate at 2–8 °C
-
Do not freeze; discard if frozen
-
After first use: May store at ≤30 °C for up to 30 days
-
Discard pen after 30 days, even if solution remains
-
Needles: Remove after each injection; dispose of safely in a sharps container
-
Keep out of sight and reach of children
What is MOUNJARO® 5mg KwikPen®
MOUNJARO® 5mg KwikPen® is a multi-dose, pre-filled pen designed to deliver 5 mg tirzepatide in 0.6 mL by subcutaneous injection once weekly. This product is indicated for the treatment of adults with type 2 diabetes mellitus and for weight management in patients who meet BMI criteria. The pen is engineered for single-patient use, providing a safe, convenient method of administration. Each device contains 2.4 mL of solution (total of 20 mg tirzepatide) and supplies four fixed doses. The solution is clear, colourless to slightly yellow, and can be injected in the abdomen, thigh, or upper arm.
-
Dose per injection: 5 mg tirzepatide in 0.6 mL
-
Total pen content: 20 mg/2.4 mL (8.33 mg/mL)
-
Number of doses per pen: 4
-
Injection sites: Abdomen, thigh, upper arm
-
Administration: Subcutaneous only
What is the Administration Protocol for MOUNJARO® 5mg KwikPen®
MOUNJARO® 5mg is administered once weekly at a consistent time, with or without meals. The dosing schedule allows for gradual escalation to minimize gastrointestinal adverse reactions.
-
Initiation: 2.5 mg weekly for 4 weeks, then increase to 5 mg
-
Titration: May increase in 2.5 mg increments every ≥4 weeks (up to 15 mg)
-
Maintenance doses: 5 mg, 10 mg, or 15 mg once weekly
-
Missed dose protocol:
-
If ≤4 days late: administer as soon as possible
-
If >4 days late: skip and resume on next scheduled day
-
-
Injection technique:
-
Attach a new sterile needle for each use
-
Dial pen until 1 appears in the window (=0.6 mL)
-
Inject subcutaneously, hold for 5 seconds until dose window shows 0
-
-
Site rotation recommended to avoid local irritation
What are the Indications for MOUNJARO® 5mg KwikPen®
MOUNJARO® is prescribed for glycaemic control in adults with type 2 diabetes mellitus and as an adjunct for weight management. Contraindications and precautions should be carefully considered in clinical use.
-
Indications:
-
Adults with type 2 diabetes as monotherapy or in combination with other antidiabetic agents
-
Adults with BMI ≥30 kg/m² (obesity)
-
Adults with BMI ≥27 to <30 kg/m² with weight-related comorbidities (e.g., hypertension, dyslipidaemia, OSA, cardiovascular disease, prediabetes)
-
-
Contraindications:
-
Hypersensitivity to tirzepatide or excipients
-
-
Precautions:
-
History of pancreatitis
-
Severe gastrointestinal disorders, such as gastroparesis
-
-
Adverse effects:
-
Common: nausea, vomiting, diarrhoea, abdominal pain, constipation
-
Risk of hypoglycaemia when combined with sulphonylurea or insulin
-
Mild to moderate injection-site reactions
-
Occasional gallbladder events
-
Rare: acute pancreatitis, hypersensitivity reactions
-
What is the composition of MOUNJARO® 5mg KwikPen®
The formulation contains tirzepatide as the active pharmaceutical ingredient, with additional excipients to ensure stability and usability.
-
Active substance: Tirzepatide 5 mg/0.6 mL
-
Total content per pen: 20 mg in 2.4 mL
-
Concentration: 8.33 mg/mL
-
Excipients: Disodium hydrogen phosphate heptahydrate, benzyl alcohol (~5.4 mg per dose), glycerol, phenol, sodium chloride, sodium hydroxide, concentrated hydrochloric acid, water for injections
-
Sodium content: <1 mmol sodium per dose (sodium-free)
What Comes in the MOUNJARO® 5mg KwikPen® Package
MOUNJARO® KwikPen® is supplied in carefully prepared packaging, but needles are not included.
-
Presentation: Pre-filled KwikPen® with 2.4 mL (4 doses)
-
Pack sizes: 1 or 3 pens per carton (availability may vary)
-
Needles: Not supplied in the package; must be obtained separately
-
Documentation: Patient Information Leaflet (PIL) and Instructions for Use (IFU) included
Does MOUNJARO® 5mg KwikPen® cause side effects?
Yes, side effects are possible, with gastrointestinal reactions being most common. These effects are usually dose-related and may decrease over time.
-
Very common: Gastrointestinal disorders – nausea, diarrhoea, vomiting, constipation, abdominal pain
-
Hypoglycaemia: Increased risk when combined with sulphonylureas or insulin; severe hypoglycaemia rare (~0.2%)
-
Injection-site reactions: Up to 3–8% of patients, generally mild
-
Gallbladder events: Reported in up to 2% of patients
-
Uncommon but serious: Acute pancreatitis
-
Rare: Severe allergic reactions (e.g., anaphylaxis, angioedema)
-
Laboratory changes: Small increases in amylase and lipase
How should MOUNJARO® 5mg KwikPen® be stored?
Strict storage conditions must be followed to maintain stability and sterility.
-
Unopened pens: Refrigerate at 2–8 °C
-
Do not freeze; discard if frozen
-
After first use: May store at ≤30 °C for up to 30 days
-
Discard pen after 30 days, even if solution remains
-
Needles: Remove after each injection; dispose of safely in a sharps container
-
Keep out of sight and reach of children