HYMOVIS® (German)
$629.00
All products are 100% genuine and sourced from trusted manufacturers, with authenticity that can be verified for complete peace of mind.
Order exactly what you need with no minimum quantity required, whether it’s a single unit or a larger order.
Your order is guaranteed to arrive safely, with full shipment tracking and dedicated support to ensure a smooth delivery experience.
Your payments are protected with industry-standard encryption and trusted payment providers, ensuring every transaction is safe, private, and secure.
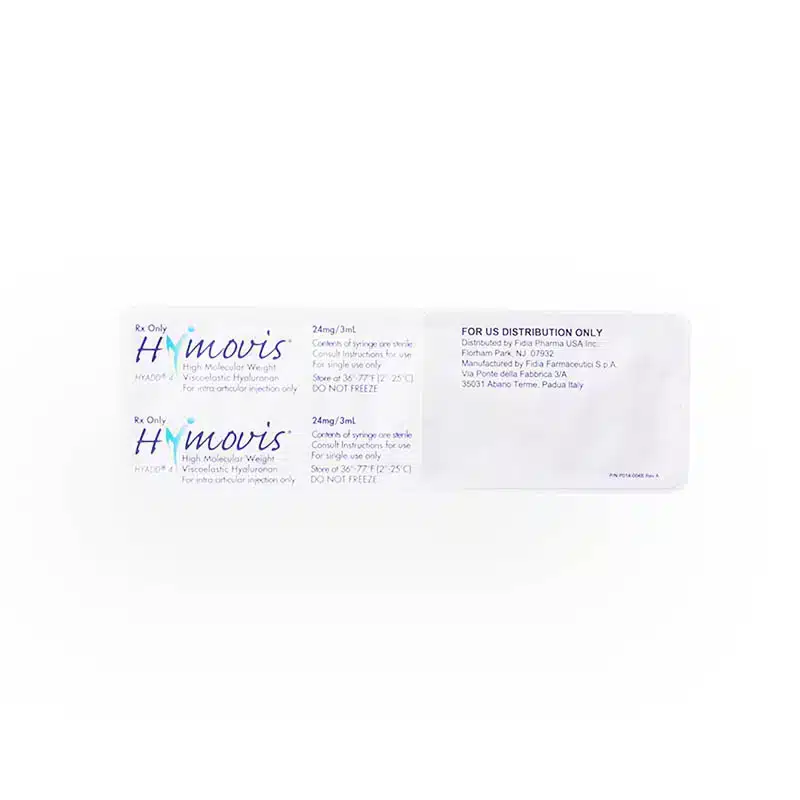
Product: HYMOVIS® (German)
Manufacturer: Fidia
Active Substance(s): HYALURONAN
Strength: 24mg/3ml
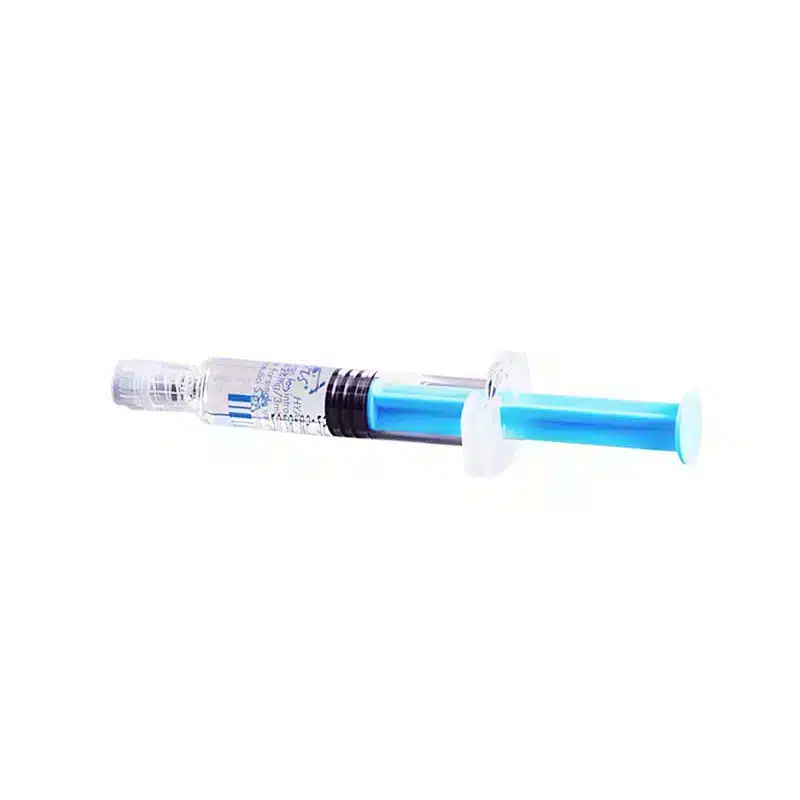
Pack Size: 2 x 5ml Pre-Filled Syringes
Accessories: Package insert
HYMOVIS® is intended to provide relief from your osteoarthritis knee pain. HYMOVIS® is a thick, gel-like solution that acts like a lubricant and shock absorber in the knee joint. HYMOVIS® injections may provide up to six months of relief from osteoarthritis knee pain.
-
Cold chain shipping available for temperature-sensitive products.
-
Orders are processed within 1–2 business days.
-
Delivery timelines vary by destination and shipping method — view our Shipping Policy for region-specific estimates.
-
Tracking information will be provided once shipped.
-
No order minimum applies.
-
If your shipment is delayed, lost, or arrives damaged, you're covered under our Refund & Replacement Policy.
We accept Visa, Mastercard, and American Express. Apple Pay and Google Pay are available upon request via your account manager.
All transactions are processed through a secure, PCI DSS–compliant system to ensure your data is fully protected.


Why Choose Medica Depot
Shop hundreds of medical products with no minimums and free shipping on large orders.
INFORMATION
WHAT IS HYMOVIS TREATMENT?
Hymovis is an injectable gel that is used to treat pain associated with knee osteoarthritis. Made of proprietary and highly modified hyaluronan that has enhanced lubricating and shock absorption abilities, Hymovis can provide long-lasting pain relief in a simple two-injection regimen. Hymovis is manufactured by Fidia USA.
HOW TO USE HYMOVIS INJECTIONS?
Hyamovis is an intra-articular injection. A thorough preprocedural consultation with a healthcare professional is essential before starting treatment. This step allows the doctor to evaluate the patient’s condition, determine the severity of arthritis, and ensure there are no contraindications. Additionally, this session provides an opportunity to discuss potential side effects, aftercare techniques, and the recommended Hymovis dosing schedule for optimal results. Once the patient is deemed suitable for treatment, a procedure date can be scheduled.
On the day of the procedure, the injection site must be properly cleaned and disinfected. Any joint effusions should be removed using an 18- to 20-gauge needle, as failure to do so may lead to undesirable side effects. The next step involves preparing the Hymovis syringe by attaching a sterile 18- to 20-gauge needle to its tip. The full 3ml of hyaluronan gel should then be injected directly into the intra-articular space without overfilling the joint. If both knees require treatment, separate syringes must be used for each joint. To complete the Hymovis dosing regimen, a second injection should be administered one week later.
After the procedure, all used needles and syringes must be disposed of in biological waste bins. It is crucial that only trained healthcare professionals perform the injections under strict aseptic conditions to ensure patient safety. Those looking to incorporate this effective arthritis treatment into their practice can buy Hymovis from reputable medical suppliers.
HOW DOES HYMOVIS TREATMENT WORK?
The flexible synovial membrane is a characteristic of synovial joints such as the hips and knees. The protective articular cartilage is a thin layer that covers the ends of bones. This protects them from friction and allows them to glide smoothly against each other. The synovial membrane also produces lubricating synovial liquid that absorbs shocks in the event of sudden impacts. The articular cartilage can become damaged from wear and tear, excess weight, or injuries. In this case, the bones will grind against each other when they move. Osteoarthritis (OA) is a condition that causes debilitating symptoms like pain, swelling, stiffness in the joints, and a lack of mobility.
Hymovis contains 8mg/ml viscoelastic, hyaluronan from biofermentation. This hyaluronan can be extracted and purified using patented technology. It is not subject to any chemical reticulating agent. The resulting hyaluronan, HYADD 4, is superior in lubricating as well as shock-absorbing properties. Hymvois is injected intraarticularly into the affected knee joints. This replaces the synovial fluid and allows patients to resume daily activities with less pain and discomfort.
HOW LONG DOES HYMOVIS?
After Hymovis treatment, patients should notice improvements in their OA within six months. Hymovis’s effectiveness is affected by the severity of the OA. Patients can get another Hymovis injection if the injected viscosupplement wears out and symptoms of OA return.
IS HYMOVIS TREATMENT SAFE?
Yes, Hymovis is a safe and effective viscosupplementation, as it is made in a strict laboratory setting. Hyaluronan, the active ingredient in Hymovis, is obtained from a strict bacterial fermentation process. After that, it undergoes a thorough purification process. Hymovis is a safe and effective intra-articular device because it does not contain any pyrogens or chemical reticulating agent. However, it is not recommended for the following patients because of its low safety profile.
- Patients allergic to hyaluronan
- Allergy to gram-positive bacterial proteins in patients
- Patients with dermal reactions (e.g. inflammations, injections, etc.) In the area or joint where you are seeking treatment.
- For more information on warnings and precautions, refer to the package insert.
WHAT ARE HYMOVIS SIDE EFFECTS?
These are some of the side effects that Hymovis can cause:
- Redness;
- Pain;
- Swelling;
- Itching or tingling sensations
- Stiffness in the joints
- To reduce side effects, tell your patients to refrain from engaging in high-impact activities such as tennis and jogging, as well as prolonged weight-bearing activities such as standing for 48 hours. However, severe reactions are still possible in some patients.
These are some of the side effects that Hymovis can cause:
- Arthropathy;
- Phlebitis;
- Tendonitis;
- Paraesthesia;
- Pruritus.
- These lists are not complete. Refer to the insert for a detailed listing of side effects.
WHAT IS HYMOVIS MADE OF?
Hymovis, a hyaluronan implant intra-articular, is intended to be injected into synovial space to relieve pain from OA. Each 5mL syringe comes preloaded with sterile Gel that contains 8mg/ml ultra-pure, high molecular weight hyaluronan. This hyaluronan has been dissolved in physiologic Saline. Hymovis should only be injected once per week in the affected knee joint.
WHAT IS HYMOVIS TREATMENT?
Hymovis is an injectable gel that is used to treat pain associated with knee osteoarthritis. Made of proprietary and highly modified hyaluronan that has enhanced lubricating and shock absorption abilities, Hymovis can provide long-lasting pain relief in a simple two-injection regimen. Hymovis is manufactured by Fidia USA.
HOW TO USE HYMOVIS INJECTIONS?
Hyamovis is an intra-articular injection. A thorough preprocedural consultation with a healthcare professional is essential before starting treatment. This step allows the doctor to evaluate the patient’s condition, determine the severity of arthritis, and ensure there are no contraindications. Additionally, this session provides an opportunity to discuss potential side effects, aftercare techniques, and the recommended Hymovis dosing schedule for optimal results. Once the patient is deemed suitable for treatment, a procedure date can be scheduled.
On the day of the procedure, the injection site must be properly cleaned and disinfected. Any joint effusions should be removed using an 18- to 20-gauge needle, as failure to do so may lead to undesirable side effects. The next step involves preparing the Hymovis syringe by attaching a sterile 18- to 20-gauge needle to its tip. The full 3ml of hyaluronan gel should then be injected directly into the intra-articular space without overfilling the joint. If both knees require treatment, separate syringes must be used for each joint. To complete the Hymovis dosing regimen, a second injection should be administered one week later.
After the procedure, all used needles and syringes must be disposed of in biological waste bins. It is crucial that only trained healthcare professionals perform the injections under strict aseptic conditions to ensure patient safety. Those looking to incorporate this effective arthritis treatment into their practice can buy Hymovis from reputable medical suppliers.
HOW DOES HYMOVIS TREATMENT WORK?
The flexible synovial membrane is a characteristic of synovial joints such as the hips and knees. The protective articular cartilage is a thin layer that covers the ends of bones. This protects them from friction and allows them to glide smoothly against each other. The synovial membrane also produces lubricating synovial liquid that absorbs shocks in the event of sudden impacts. The articular cartilage can become damaged from wear and tear, excess weight, or injuries. In this case, the bones will grind against each other when they move. Osteoarthritis (OA) is a condition that causes debilitating symptoms like pain, swelling, stiffness in the joints, and a lack of mobility.
Hymovis contains 8mg/ml viscoelastic, hyaluronan from biofermentation. This hyaluronan can be extracted and purified using patented technology. It is not subject to any chemical reticulating agent. The resulting hyaluronan, HYADD 4, is superior in lubricating as well as shock-absorbing properties. Hymvois is injected intraarticularly into the affected knee joints. This replaces the synovial fluid and allows patients to resume daily activities with less pain and discomfort.
HOW LONG DOES HYMOVIS?
After Hymovis treatment, patients should notice improvements in their OA within six months. Hymovis’s effectiveness is affected by the severity of the OA. Patients can get another Hymovis injection if the injected viscosupplement wears out and symptoms of OA return.
IS HYMOVIS TREATMENT SAFE?
Yes, Hymovis is a safe and effective viscosupplementation, as it is made in a strict laboratory setting. Hyaluronan, the active ingredient in Hymovis, is obtained from a strict bacterial fermentation process. After that, it undergoes a thorough purification process. Hymovis is a safe and effective intra-articular device because it does not contain any pyrogens or chemical reticulating agent. However, it is not recommended for the following patients because of its low safety profile.
- Patients allergic to hyaluronan
- Allergy to gram-positive bacterial proteins in patients
- Patients with dermal reactions (e.g. inflammations, injections, etc.) In the area or joint where you are seeking treatment.
- For more information on warnings and precautions, refer to the package insert.
WHAT ARE HYMOVIS SIDE EFFECTS?
These are some of the side effects that Hymovis can cause:
- Redness;
- Pain;
- Swelling;
- Itching or tingling sensations
- Stiffness in the joints
- To reduce side effects, tell your patients to refrain from engaging in high-impact activities such as tennis and jogging, as well as prolonged weight-bearing activities such as standing for 48 hours. However, severe reactions are still possible in some patients.
These are some of the side effects that Hymovis can cause:
- Arthropathy;
- Phlebitis;
- Tendonitis;
- Paraesthesia;
- Pruritus.
- These lists are not complete. Refer to the insert for a detailed listing of side effects.
WHAT IS HYMOVIS MADE OF?
Hymovis, a hyaluronan implant intra-articular, is intended to be injected into synovial space to relieve pain from OA. Each 5mL syringe comes preloaded with sterile Gel that contains 8mg/ml ultra-pure, high molecular weight hyaluronan. This hyaluronan has been dissolved in physiologic Saline. Hymovis should only be injected once per week in the affected knee joint.